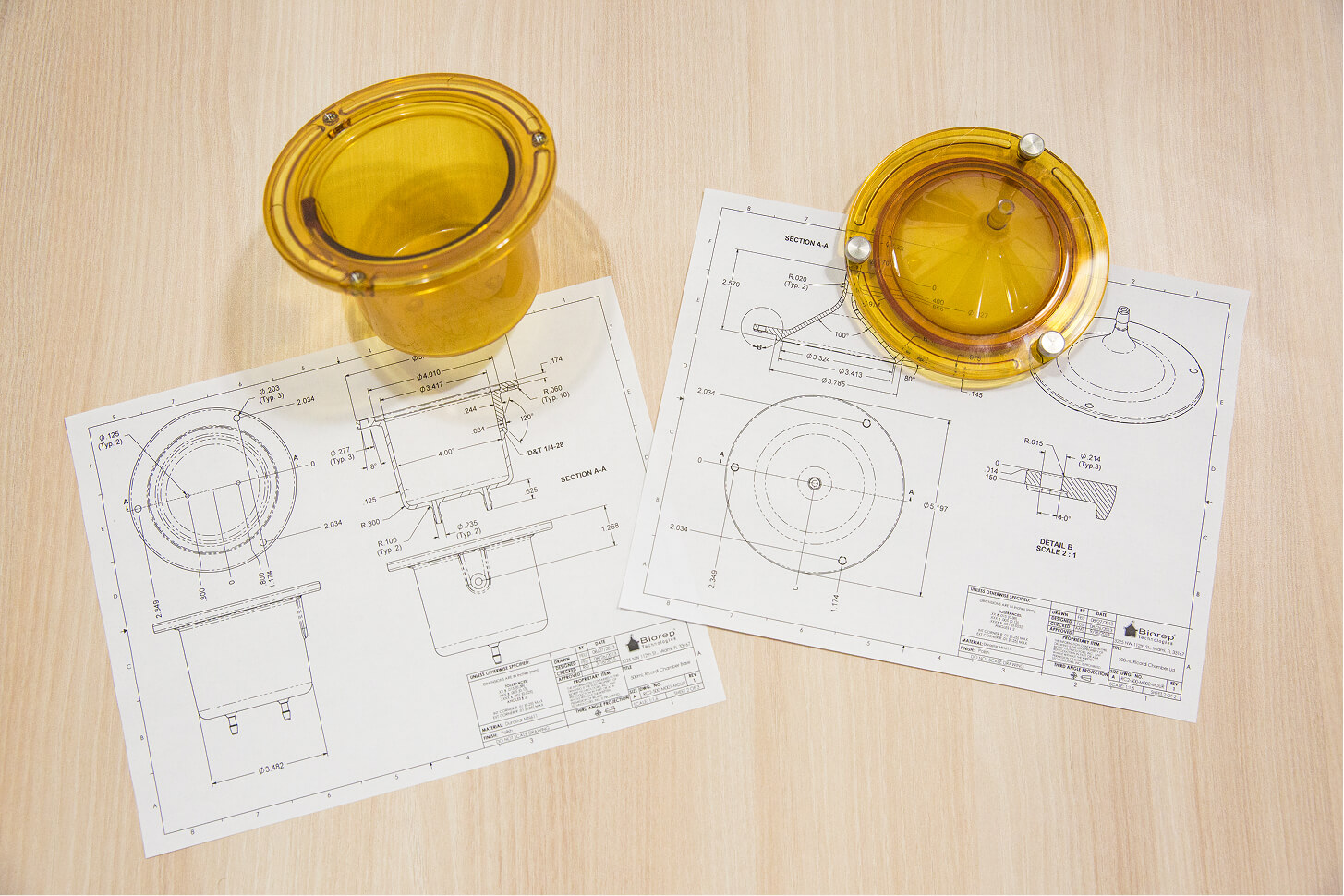
Validate form, fit, functionality, manufacturability, and user interface features, fast. Biorep’s rapid prototyping capabilities include quick iteration, design for manufacturability (DFM) review, tolerancing, and early risk reduction.
We take an iterative approach to ensure prototypes progressively mature into manufacturable designs. This helps de-risk the design in later stages.