At Biorep Technologies, Inc., we don’t just build components, we bring your device to life. When you engage our medical device assembly services, you’re teaming up with a partner who understands that even the smallest actuator or circuit board can make a world of difference to patient outcomes.
MEDICAL DEVICE ASSEMBLY SERVICES
Seamless Precision: Your Partner for Medical Device Assembly Services
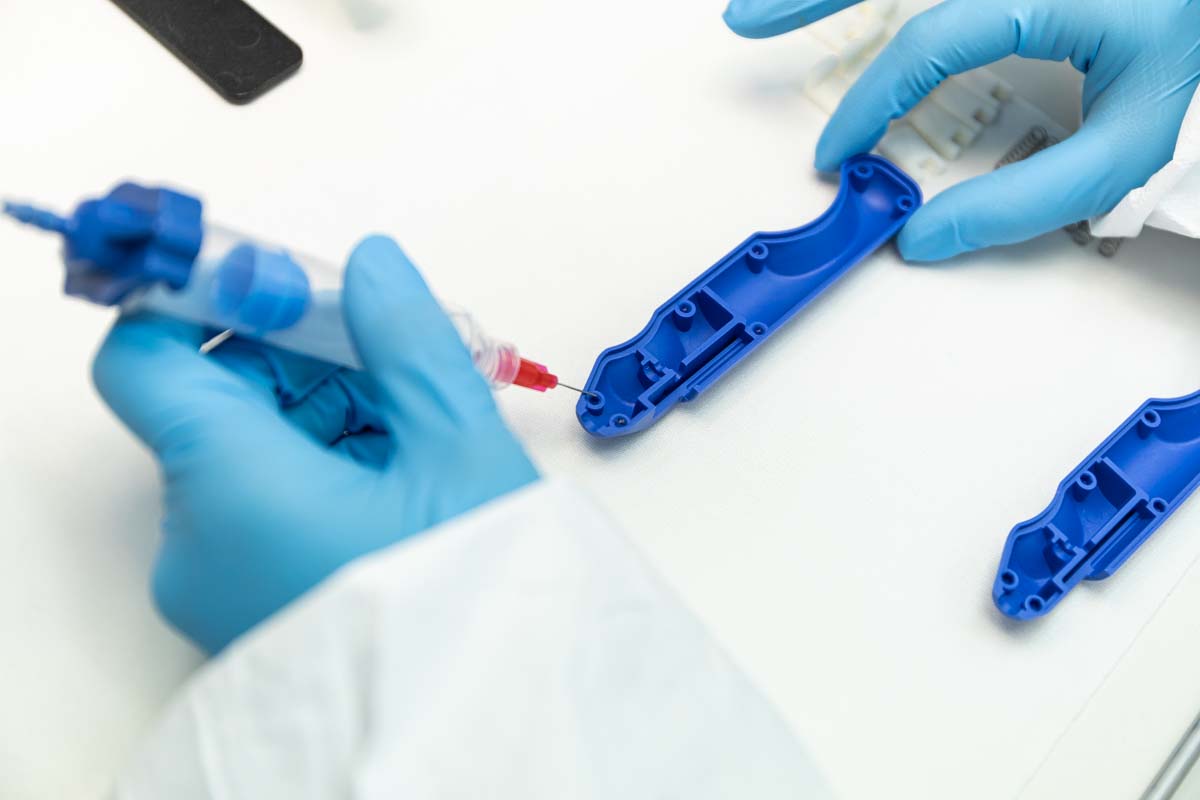
Why Assembly Matters (and Why We Do It Differently)
Outsourcing assembly is more than finding a bench and a technician, it’s about combining engineering excellence, regulatory discipline, and controlled manufacturing environments for supply chain agility. We’ve been exclusively focused on regulated medical devices for over 30 years. When you bring us your project, you’re tapping into a system built around quality from the ground up including design, prototyping, manufacturing, and yes, assembly.
End-to-End Assembly: From Micro-Mechanical to Full System Delivery
Under our roof, our team handles complex subassemblies, full device integration, validated packaging and quality in-process and final inspections. Our medical device assembly services include:
Including mechanical, micro-mechanical and electromechanical.
Utilizing both automated and manual processes when either volume demands and or precision matters most.
In-process inspections, functional and environmental testing to ensure every unit shipped meets your specification.
We create regulatory complaint labelling and validate your packaging configurations to ensure your packaging can withstand environmental and shipping conditions and maintain sterility when applicable.
This means our clients benefit from fewer hand-offs, faster turnaround, and tighter control across the supply chain.
Cleanroom Discipline: Exacting Standards for High-Stakes Devices
Many medical devices demand an environment free from contaminants. We’ve designed our facility to support cleanroom assembly, including environments suitable for ISO 8 cleanroom assembly practices (or equivalent controlled environments) to manage particulate, microbial, and environmental risk.
By integrating this cleanroom discipline into our assembly workflows, we provide you with a foundation for reliability, which is ideal when your device is destined for surgical theatres, implantable use, or sensitive diagnostics.
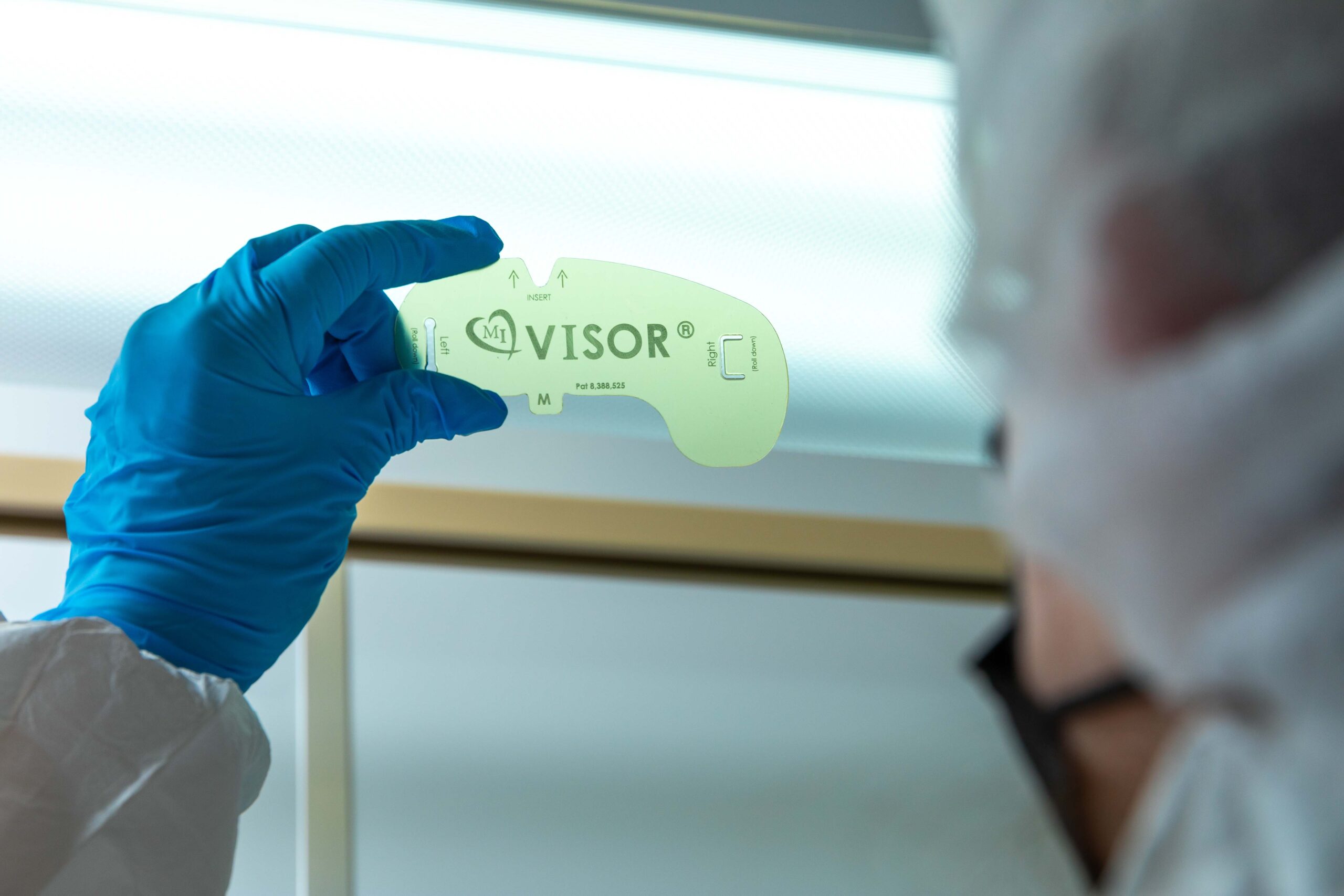

Sterile-Device Manufacturing: Your Pathway to Launch
When your device must leave the line ready for sterile packaging, we’re already ahead of the curve. Our capability in sterile device manufacturing means we incorporate sterilization-compatible processes, packaging interfaces, labeling controls, and pre-sterile release inspections into the assembly line.
Because we also cover full contract manufacturing, design, and prototyping, our execution is not an afterthought, but part of the device’s DNA.
If your device must arrive in a sterile pack, with traceability, validated processes, and global market readiness, you’re talking to the right partner: Biorep.
Electronic-Integration Services: More Than Just Mechanical Assembly
In today’s med-tech world, devices are rarely “just mechanical”.
They can entail embedded sensors, electronics, firmware, connectivity, user-interfaces, which means your assembly partner must be able to handle a wide range of complex assembly needs.
Our electronic integration services mean we can take printed-circuit boards (PCBs), flex circuits, sensor modules, actuators, power systems, connectivity modules and integrate them into your device.
Because we conduct assembly in-house at Biorep, you benefit from tighter control, quicker iterations, and higher responsiveness to change. We bring the integration discipline you need.


Our Value Proposition: Partner, Not Vendor
Choosing Biorep for your medical device assembly services means more than “let’s get this built.” It means:
Domain focus
We only do medical devices, so our systems, culture, and expertise are aligned with regulated-device realities.
Reduced vendor count
When you let us prototype, manufacture, and assemble, you can maintain one point of contact, simplify oversight, and reduce risk.
Scalable capacity
From low-volume pilot builds in our cleanroom to full commercial production, we scale alongside your program.
Global-ready
Our quality systems and regulatory infrastructure (ISO 13485 certified, FDA-registered) support global med-tech leaders.
How We Engage: Your Roadmap to Success
When you come to us for medical device assembly services, the journey looks like this:
01
We map technical challenges, regulatory context, and your business roadmap so assembly isn’t a surprise checkpoint, it’s part of the design strategy.
02
Early builds validate your subsystem integrations (mechanical, electromechanical, electronics). We work to be proactive prior to assembly to preempt issues and potential mismatches.
03
We build the tools, fixtures, layout, inspection flows, and incorporate change control, so when we manufacture your assembly, it is ready for scale.
04
With validated processes in hand, we shift into production mode, so your device assemblies are shipped, monitored, and traceable.
05
Post-launch, we manage component obsolescence, design changes, supply-chain shifts because the work doesn’t end at initial product launch.
Ready When You Are
If you’re developing a device where assembly counts: where mechanical accuracy meets electronics, where sterility meets cleanroom controls, where global markets demand unwavering quality, then our medical device assembly services are ready to accelerate your launch.
Reach out. Let’s schedule a discovery workshop. Share your design, your challenges, and your volume targets. We’ll map your needs, propose a phased plan, and deliver the assembly capability your device deserves. At Biorep, we’re not just contracting your assembly, we’re partnering in the success of your device.



